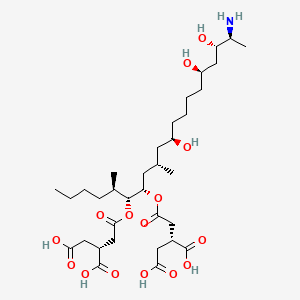
fumonisin b1
fumonisin b1 is a lipid of Sphingolipids (SP) class. Fumonisin b1 is associated with abnormalities such as Infection, Kidney Diseases, Liver diseases, DERMATITIS HERPETIFORMIS, FAMILIAL and Malnutrition. The involved functions are known as Gene Expression, Anabolism, Signal, Biosynthetic Pathways and Regulation. Fumonisin b1 often locates in Body tissue, Microsomes, microsomal membrane, Protoplasm and Mitochondria. The associated genes with fumonisin b1 are Genome, P4HTM gene, FATE1 gene, BCL2 gene and TMEM132D gene. The related lipids are dihydroceramide, ceramide 1-phosphate, Sphingolipids, Fatty Acids and Palmitates.
References related to functions published in J. Biol. Chem.
| PMID | Journal | Published Date | Author | Title |
|---|---|---|---|---|
| 8662871 | J. Biol. Chem. | 1996 | Hidari KIPJ S et al. | Complete removal of sphingolipids from the plasma membrane disrupts cell to substratum adhesion of mouse melanoma cells. |
| 9006952 | J. Biol. Chem. | 1997 | de Chaves EI et al. | Elevation of ceramide within distal neurites inhibits neurite growth in cultured rat sympathetic neurons. |
| 7768956 | J. Biol. Chem. | 1995 | Strum JC et al. | Ceramide triggers meiotic cell cycle progression in Xenopus oocytes. A potential mediator of progesterone-induced maturation. |
| 10364237 | J. Biol. Chem. | 1999 | Liao WC et al. | Ataxia telangiectasia-mutated gene product inhibits DNA damage-induced apoptosis via ceramide synthase. |
| 11801602 | J. Biol. Chem. | 2002 | Chalfant CE et al. | De novo ceramide regulates the alternative splicing of caspase 9 and Bcl-x in A549 lung adenocarcinoma cells. Dependence on protein phosphatase-1. |
| 12200446 | J. Biol. Chem. | 2002 | Jenkins GM et al. | Acute activation of de novo sphingolipid biosynthesis upon heat shock causes an accumulation of ceramide and subsequent dephosphorylation of SR proteins. |
| 12424251 | J. Biol. Chem. | 2003 | Hinkovska-Galcheva V et al. | Enhanced phagocytosis through inhibition of de novo ceramide synthesis. |
| 12912983 | J. Biol. Chem. | 2003 | Riebeling C et al. | Two mammalian longevity assurance gene (LAG1) family members, trh1 and trh4, regulate dihydroceramide synthesis using different fatty acyl-CoA donors. |
| 10702247 | J. Biol. Chem. | 2000 | Mao C et al. | Cloning of an alkaline ceramidase from Saccharomyces cerevisiae. An enzyme with reverse (CoA-independent) ceramide synthase activity. |
| 15180992 | J. Biol. Chem. | 2004 | Le Stunff H et al. | Role of sphingosine-1-phosphate phosphatase 1 in epidermal growth factor-induced chemotaxis. |
| 21388949 | J. Biol. Chem. | 2011 | Mullen TD et al. | Ceramide synthase-dependent ceramide generation and programmed cell death: involvement of salvage pathway in regulating postmitochondrial events. |
| 19095642 | J. Biol. Chem. | 2009 | Zitomer NC et al. | Ceramide synthase inhibition by fumonisin B1 causes accumulation of 1-deoxysphinganine: a novel category of bioactive 1-deoxysphingoid bases and 1-deoxydihydroceramides biosynthesized by mammalian cell lines and animals. |
| 20097765 | J. Biol. Chem. | 2010 | Penno A et al. | Hereditary sensory neuropathy type 1 is caused by the accumulation of two neurotoxic sphingolipids. |
| 21926169 | J. Biol. Chem. | 2011 | Kim WY et al. | Inhibitor of apoptosis (IAP)-like protein lacks a baculovirus IAP repeat (BIR) domain and attenuates cell death in plant and animal systems. |
| 17504762 | J. Biol. Chem. | 2007 | Kitatani K et al. | Mechanism of inhibition of sequestration of protein kinase C alpha/betaII by ceramide. Roles of ceramide-activated protein phosphatases and phosphorylation/dephosphorylation of protein kinase C alpha/betaII on threonine 638/641. |
| 15546881 | J. Biol. Chem. | 2005 | Becker KP et al. | Selective inhibition of juxtanuclear translocation of protein kinase C betaII by a negative feedback mechanism involving ceramide formed from the salvage pathway. |
| 16043487 | J. Biol. Chem. | 2005 | Kuroyanagi M et al. | Vacuolar processing enzyme is essential for mycotoxin-induced cell death in Arabidopsis thaliana. |
| 18625703 | J. Biol. Chem. | 2008 | Nieto FL et al. | Sphingolipid metabolism is a crucial determinant of cellular fate in nonstimulated proliferating Madin-Darby canine kidney (MDCK) cells. |
| 17895250 | J. Biol. Chem. | 2007 | Le Stunff H et al. | Recycling of sphingosine is regulated by the concerted actions of sphingosine-1-phosphate phosphohydrolase 1 and sphingosine kinase 2. |